Alcohol Synthesis Problems
Extra Problems
7. Draw the following compounds:
a) isopropyl magnesium bromide
b) 3-heptanol
c) cyclohexanol
d) 4-iodophenol
e) 3-cyclopenten-1-ol
f) p-chlorophenol
g) (R)-pentane-1,4-diol
h) cyclohexane-1,2,3-triol
i) o-bromophenol
j) propylene glycol
k) (Z)-4-chloro-3-buten-2-ol
l) 2-methylbut-3-yn-1-ol
m) m-cresol
n) cyclohexylmethanol
o) 4-bromobutan-1-ol
p) propyl mercaptan
q) pentanethiol
r) butyl mercaptan
s) butane-2-thiol
8. Name the following compounds:
a)
b)

c)
d)

e)

f)
g)
h)
i)

j)

k)

l)

9. A little chemistry joke. . . What words do the following structures represent? Think ortho, meta, and para.

10. Which of the following IR spectra is that of an alcohol?

11. The following alcohol can be synthesized from two different alkenes. Draw these two alkenes and then give the reagents you would use to synthesize the alcohol from each of these alkenes.

12. Draw the products of the following reactions.
a) 1-methylcyclopentene + dilute aqueous acid
b) 1-methylcyclopentene + aqueous Hg(OAc)2 followed by NaBH4
c) 1-methylcyclopentene + a cold, dilute, neutral solution of potassium permanganate
d) 1-methylcyclopentene + BH3.THF followed by H2O2 and NaOH
e) 1-methylcyclopentene + OsO4 followed by H2O2
f) 1-methylcyclopentene + peroxybenzoic acid in water
g) 2-methylpropene + aqueous Hg(OAc)2 followed by NaBH4
h) 2-methylpropene + dilute aqueous H2SO4
i) 2-methylpropene + BH3.THF followed by H2O2 and NaOH
j) 2-methylpropene + OsO4 followed by H2O2
k) 2-methylpropene + MCPBA followed by acid/water
l) cyclopentene + peroxybenzoic acid in water
m) cyclopentene + a cold, dilute, neutral solution of potassium permanganate
n) cyclopentene + OsO4 followed by H2O2
o) cyclopentene + MCPBA followed by acid/water
13. Show how you would prepare the following compounds starting with an alkene and any other reagents you need.
a) 1-heptanol
b) 2-heptanol
c) 1-ethyl-1-cyclopentanol
d) 2-ethyl-1-cyclopentanol
14. How could you make 2-methylcyclopentanol from 1-bromo-1-methylcyclopentane? More than one reaction is required.
15. Determine what aldehyde, ketone, or epoxide would react with ethyl magnesium bromide such that, upon quenching with aqueous acid, would make the following compounds.
a)

b)

c)

d)

e)

f)

g)

h)
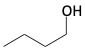
16. Determine an appropriate Grignard reagent that would react with acetaldehyde (CH3CHO) such that, upon quenching with aqueous acid, would make the following compounds.
a)

b)

c)

d)

e)

17. Determine an appropriate Grignard reagent that would react with 1,2-epoxypropane such that, upon quenching with aqueous acid, would make the following compounds.

1,2-epoxypropane
a)

b)

c)


d)
e)

18. Draw the structure of an acid chloride that would react with an excess of methyl magnesium bromide such that, upon quenching with aqueous acid, would make the following compounds. Then, draw an appropriate ester for each problem that you could use instead of the acid chloride.
a)

b)

c)

d)

e)

19. Give two ways you could synthesize the following compounds using the reaction of a Grignard reagent and an aldehyde.
a)

b)

c)

d)

20. Give three ways you could synthesize the following compounds using the reaction of a Grignard reagent and a ketone.
a)

b)

c)

d)

21. For each of the following compounds, show how it could be synthesized from an acid chloride or ester and an excess of a Grignard reagent.
a)

b)

c)

d)

22. Reducing a carbonyl compound can make the following compounds. Show what carbonyl compound and appropriate reducing agent can be used to make each of the following.
a)

b)

c)

d)

e)
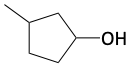
23. Draw the products for each of the following reactions.
a)

b)
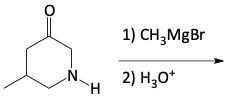
c)

d)

e)

f)

g)

h)

i)

j)


k)
l)

m)

n)

o)

p)

q) H2C=CHCO2H + 1) LiAlH4, 2) acid/water
r) CH3CH2COCl + 1) LiAlH4, 2) acid/water
s) CH3CO2CH2CH2CHO + 1) NaBH4, 2) acid/water
t) CH3COCH2CH=CHCH2CO2CH3 + 1) LiAlH4, 2) acid/water
u) PhCH2CHO + 1) LAH, 2) acid/water
v) iodohexane + sodium hydrosulfide
24. What reagents are needed to perform the following transformations?
a) CH3CO2CH2CH2CHO to CH3CO2CH2CH2CH2OH
b) CH3CH2CO2H to CH3CH2CH2OH
c) CH3CH2CH=CHCH2CHO to CH3CH2CH2CH2CH2CH2OH
d) CH3CO2CH3 to CH3CH2OH
e)

f)

g)

h)

i)
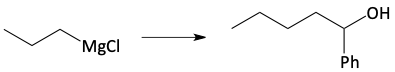
j)

k)

l)

m)


n)
o)

p)

q) CH3CH2CH2CO2H to CH3CH2CH2CH2OH
r) PhCO2Cl to Ph3COH
s) PhCO2Cl to PhCH2OH
t) HCO2CH2CH3 to (Ph)2CHOH
u) 1,2-epoxybutane to 3-hexanol
v) PhCH2Cl to PhCH2CH2OH
w) 1-bromobutane to 1-pentanol
x) EtCO2CH3 to (Et)3COH
y) 1-bromocyclopentane to 1-cyclopentyloctane
z) propene to propylene glycol
aa) bromobenzene to 2-phenylethanol
bb) bromopentane to pentanethiol
25. A mixture of ortho, meta, and para aromatic compounds is below.

a) Label A, B, and C as ortho-, meta-, or para-.
b) What would be the product of the reaction of A with an excess of NaBH4 followed by hydrolysis?
c) What would be the product of the reaction of C with an excess of LiAlH4 followed by hydrolysis?
d) Draw the product and the mechanism of the reaction of B with an excess of ethyl grignard (CH3CH2MgBr) followed by hydrolysis.
26. The following reactions did not work as planned. What is wrong with each of these reactions?
a)

b)

c)

d)
