New Syntheses of alcohols
More on the nucleophilic attack of carbonyls
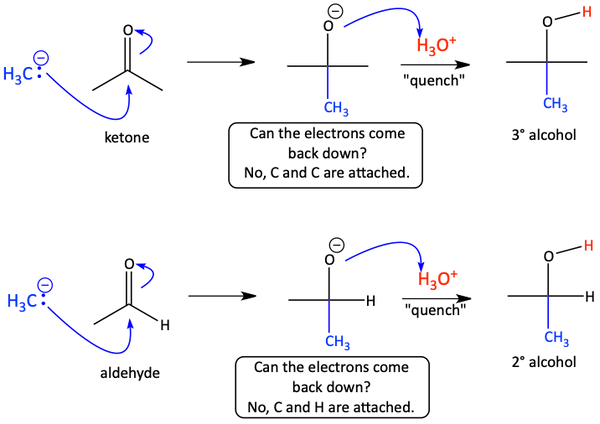
Let’s pause for a moment to more fully think about nucleophiles attacking carbonyl compounds. When a nucleophile, like acetylide or some other negatively charged species, attacks the partially positive carbon atom of the carbonyl. The electrons of the carbonyl pi bond go up on to the oxygen atom, giving it a formal negative charge. Every time this happens, a question needs to be asked. “Can the electrons come back down?”
What we are really asking with this question is whether the groups attached to the carbonyl are good leaving groups or not. If one of the groups is a good leaving group, like Cl or OR, the electrons come back down to reform the carbonyl pi bond. This would make five bonds to the carbonyl carbon, so the leaving group must leave. Acid chlorides and esters have good leaving groups on the carbonyl because Cl and O like the negative charge when they leave. Whenever the electrons come back down, a carbonyl compound is reformed. When a carbonyl is reformed, another nucleophile can attack, and we ask the same question again. Usually, at this point, the electrons cannot come back down because the remaining groups are all poor leaving groups.
Aldehydes and ketones do not have good leaving groups on the carbonyl carbon because H and alkyl groups are terrible leaving groups because H and C are not very electronegative and are too small to want the negative charge. In the case where there is no good leaving group on the carbonyl, the electrons stay up on the carbonyl oxygen as a negative charge.
Finally, a source of H+, protons, must be added to the reaction. This is called quenching the reaction. Typically, a dilute aqueous acid is used as the proton source.
Closely study the following scheme to make sure you understand how the nucleophilic attack of a carbonyl works. You’ll find knowing this process to be very helpful in understanding the reactions of carbonyls with acetylides, Grignard reagents, and reducing agents.

Grignard and Organolithium Reagents
Organic chemistry is the chemistry of carbon-based compounds. But, surprisingly, so far, we have seen very few reactions that form carbon-carbon bonds. But, one great way to do so is to use Grignard (pronounced grin-yard) reagents. Grignard reagents, and the Grignard reaction, were discovered by Victor Grignard. Grignard was awarded the 1912 Nobel Prize in Chemistry for this discovery.
Much like acetylides, Grignard reagents have a negatively charged carbon atom. This negatively charged carbon atom makes it a good nucleophile. Grignard reagents are made by reacting alkyl halides with magnesium. Usually, a solvent like ether is used.

The magnesium metal in a Grignard reagent, like other metals, carries with it a positive charge. Therefore, the carbon atom attached to the Grignard reagent has a negative charge. Grignard reagents can be thought of as an R group with a lone pair of electrons and a negative formal charge. Because of the localized, small negative charge, Grignard reagents are aggressive nucleophiles.

A reagent that is very similar to a Grignard reagent is an alkyl lithium or an organolithium reagent. These also behave as carbon species that contain a negative charge. They are prepared by the reaction of an alkyl halide with lithium.

Organolithium reagents, with their positive lithium metal, also have a negative carbon atom. They react like Grignard reagents, like negative carbon species. This is how you should think about them.

Let’s look at the attack of methyl Grignard on various carbonyl compounds. As you study these, make sure each step of the mechanism makes sense to you. Learn what questions you should ask yourself as you do these mechanisms.
Grignard attack of aldehydes and ketones
The carbanion of the Grignard reagent attacks the carbon atom of the carbonyl because the carbon atom has a slight positive charge compared to the more electronegative oxygen atom. The electrons go up onto the oxygen atom. When this happens, we ask ourselves, “Can the electrons come back down?” The answer to that question for aldehydes and ketones is “No” since alkyl groups and hydrogen atoms are poor leaving groups. The negative charge stays on the oxygen atom until it is quenched with acid and water to make an alcohol. For ketones, you can notice that a tertiary alcohol is formed. For aldehydes, a secondary alcohol is formed.
Grignard reagents reacting with an aldehyde or ketone
Grignard attack of esters and acid chlorides
For esters and acid chlorides, the carbanion of the Grignard reagent again attacks the carbon atom of the carbonyl because the carbon atom has a slight positive charge compared to the more electronegative oxygen atom. The electrons go up onto the oxygen atom. When this happens, we ask ourselves, “Can the electrons come back down?” The answer to that question for aldehydes and ketones is “Yes” since chloride and -O-R are pretty decent leaving groups. Therefore, the carbonyl can be reformed. The carbonyl can be attacked again by the Grignard reagent. The electrons go back up on the oxygen atom. Can they come back down? The answer is “No” because only alkyl groups remain, and they are poor leaving groups. The negative charge stays on the oxygen atom until it is quenched with acid and water to make an alcohol. Tertiary alcohols are then formed.

Grignard reagents reacting with an ester

Grignard reagents reacting with an acid chloride
Grignard reagents not only react with the carbonyl functional group, but they react with other pi bonds between two atoms of differing electronegativities. Alkenes, with a pi bond between two carbon atoms, is not attacked because there is no difference in electronegativities. Neither carbon atom has a partial positive charge. The following can also be attacked by Grignard reagents.

Non-carbonyl functional groups that react with Grignard reagents
Solvent for Grignard reagent
One major drawback to Grignard reagents is that they are so negative and such reactive nucleophiles, if any type of acidic hydrogen is around, the Grignard reagent will immediately grab the H+ proton and “quench” itself. It will become an alkane and not attack a carbonyl. These protons are sometimes hidden, so we need to be on the watch for them. Anytime you see a hydrogen atom on an oxygen atom, a nitrogen atom, a sulfur atom, or a terminal acetylene, it is bad news for Grignard reagents. Hydrogen atoms on carbon atoms are no problem. They are attached to carbon atoms too well.
Hydrogen atoms that protonate Grignard reagents

What happens when a Grignard reagent encounters an acid proton
This means that we need to be careful what types of solvents we can use for a Grignard reaction. Alcohols and water are no good as they contain acidic hydrogen atoms on oxygen atoms. Grignard reactions are usually run in diethyl ether. The ether needs to be specially prepared. It must be a dry solvent. That means that all traces of water are removed from the ether solvent.
Reaction of a Grignard reagent with a carboxylic acid
Look at this theoretical reaction of a Grignard reagent with a carboxylic acid. Do you foresee any problem with this reaction?

From what we just learned; the Grignard reagent will attack the proton on the oxygen making an alkane. This will effectively kill what we were trying to do in this reaction. The carbonyl will not be attacked.

Grignard reaction with epoxides
The reaction of a Grignard reagent with an epoxide is a triangle reaction. The aggressive, nucleophilic, Grignard reagents act like a strong criminal in the triangle reaction and the epoxide is a neutral victim. The strong criminal, the aggressive Grignard reagent, will attack the least crowded, easiest to get to side of the epoxide. The ring opens up to make an alkoxide (alkyl group with a negative oxygen). The alkoxide can then be quenched with acid and water to form an alcohol.

Grignard reagent reacting with an epoxide
Notice, two carbon atoms are between the CH3 from the group added from the Grignard reagent and the hydroxyl group of the alcohol. This is because there are two carbon atoms in an epoxide. In all of the reactions of a Grignard reagent with a carbonyl, there is only one carbon atom between the group added from the Grignard reagent and the hydroxyl group.
