Introduction
Alcohols are compounds that contain the hydroxyl (-OH) functional group. Alcohols can be classified as primary, secondary, or tertiary. Their classification is determined by how many carbon atoms are attached to the carbinol carbon atom (the one attached to the hydroxyl group). If only hydrogen atoms are attached to the carbinol carbon atom, it is methanol (methyl alcohol). If one carbon atom is attached, it is a primary alcohol. If two carbon atoms are attached, it is a secondary alcohol. If three carbon atoms are attached, it is a tertiary alcohol. If a phenyl group is attached to the hydroxyl group, it is classified as a phenol.

Classification of alcohols
Nomenclature
IUPAC Names
To name an alcohol, the longest alkane chain is identified. The –e suffix is removed and is replaced with an –ol suffix.

If the hydroxyl group is not on the end, #1, carbon atom, a number needs to be used to identify the location of the hydroxyl group. The carbon chain should be numbered starting at the end closest to the hydroxyl group.

If we need to treat an alcohol, or hydroxyl group, as a substituent because it is attached to a more important part of the molecule, it is named “hydroxy”. Let’s look at the following molecule as an example. Cyclopentanol is the main part of the molecule. The ring is numbered starting at the carbinol carbon atom (in black). The substituent is on carbon #3 of the ring and is itself numbered (in blue) starting closest to the main part of the molecule, the ring. This puts the hydroxyl group on the 2nd carbon atom of the substituent.

Sometimes, it is difficult to know which way to number a chain because two different functional groups are equally distant from the two different ends. Take for example this alkene/alcohol. On the left molecule, we see that the hydroxyl group is two carbons away from an end. On the molecule on the right, we see that the alkene is between carbons 2 and 3 and would get the number 2 associated with it. So, which is preferred?

IUPAC has assigned a priority for various functional groups when a situation like this occurs. I wouldn’t waste my time memorizing every detail of this list, but you should know it exists if you need to make nomenclature priority decisions.
High Priority
Carboxylic acids
Esters
Aldehydes
Ketones
Alcohols
Amines
Alkenes
Alkynes
Alkanes
Ethers
Halides
Low Priority
Priority of functional groups in IUPAC naming
But, you should recognize that in general, the more oxidized a functional group is (more carbon-oxygen bonds), the higher its priority. So, the hydroxyl group (with one C-O bond) is higher priority than the alkene (zero C-O bonds). We therefore assign the hydroxyl group the lowest number and find that the correct name for this molecule is:

A hydroxyl group that is being named as a substituent attached to the main part of the molecule is called “hydroxy”. In the following molecule, the cyclopentanol is the main group. When numbering the ring, the carbinol carbon atom is numbered 1. The ethyl/alcohol substituent at carbon 3 of the ring is numbered starting closest to the ring. There are two carbon atoms, so it is called “ethyl”. The hydroxyl group is on carbon 2 of the ethyl substituent giving the 2-hydroxyethyl substituent name.

Diols, compounds with two hydroxyl groups, are named by finding the longest carbon chain that includes the two hydroxyl groups. The chain is numbered to give one of the alcohols the lowest number possible. If there are two hydroxyl groups, the suffix diol is used. If there are three hydroxyl groups, triol is used, etc.

Common Names
Alkyl alcohols
Alcohols are commonly named with the name of the alkyl chain followed by the word “alcohol”.

Vicinal Diols
Vicinal diols, two hydroxyl groups on neighboring carbon atoms, are called glycols. You’ll recall that glycols can be formed from the reaction of an alkene with osmium tetroxide (OsO4), potassium permanganate (KMnO4), or epoxidation followed by acid/water. Glycols are commonly named by first giving the common name of the alkene from which the glycol is made followed by the term “glycol”.

Phenols
A phenol is a hydroxyl group (alcohol) attached to a phenyl ring.

The following phenols have a chlorine group attached and are called chlorophenols. When numbering them, the alcohol is higher priority than the halide, so the carbinol carbon atom is numbered 1. The chloro groups are then assigned the appropriate number.

When two groups are attached to a benzene ring, there is a common way to name them using the prefixes ortho, meta, and para. The way to remember the order of the prefixes is to remember “ROMP”. Let’s look at a generic R group on a phenyl ring. The carbon atom attached to the R group is called the ipso carbon atom. The carbon atom next door to the R group, the 2nd carbon atom, is the ortho carbon atom. The 3rd carbon atom is meta, and the 4th carbon atom is para. This spells “ROMP” around the ring.

ortho, meta, para positions in disubstituted benzenes
The chlorophenols from above have the following common names as well:

Methylphenols are commonly called cresols. So, a benzene ring that contains both a methyl group and a hydroxyl group can be called ortho, meta, or para cresol depending on the location of the two groups.

1. Name the following compounds.
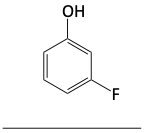



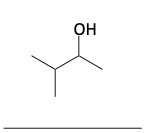

Thiols
Sulfur is directly below oxygen in the periodic table. Being in the same column, two have the same number of valence electrons and share some chemical properties. Sulfur is a little larger and less electronegative than oxygen. Thiols are the sulfur analogues of alcohols.
Thiol
Nomenclature
IUPAC
Thiols are named with the alkane plus the suffix “thiol”.

Common
Thiols are sometimes called mercaptans. For instance, CH3-SH has the common name of methyl mercaptan.
Examples
Many thiols stink. Thiols are responsible for the stink of skunks, oysters, garlic, and onions. This is a list of some common thiols.

Preparation
Thiols can be prepared from the reaction of sodium hydrosulfide with an alkyl halide via an SN2 reaction.

Properties of simple alcohols
Methanol
Methanol, or methyl alcohol, is a colorless liquid. It is a good fuel. Since 1965, it has been used in the Indianapolis racing cars. The reason it is used is it is easier to put out a fire with water if methanol is the fuel instead of gasoline. Methanol can also be made by heating wood in the absence of air. This is why methanol is sometimes called “wood alcohol”.
Methanol is a good solvent. It is used in paints and varnishes. It is dangerous to drink because the liver would oxidize it into formaldehyde and formic acid. Two tablespoons of methanol can kill somebody. We will discuss this more in the next chapter.

Ethanol
Ethanol, or ethyl alcohol, is the alcohol in booze. It is sometimes called “drinking alcohol” or “grain alcohol”. It is called grain alcohol because it is produced by the yeast fermentation of sugars from plants or grains. Ethanol usually has a little bit of water in it. To try to make ethanol have less water, to be purer and more potent, it can be distilled. But, the best a distillation can produce is 95% ethanol. In order to get 100% ethanol, or absolute ethanol, chemical drying agents must be used. Since people can drink alcohol, the government taxes it. So, in the chemistry lab, these extra taxes are costly if we simply want to use ethanol as a solvent. Therefore, we can buy “denatured” alcohol. Denatured alcohol means the alcohol purchased is poisoned. Typically, a little methanol is added to the ethanol to make it unfit to drink. So, DO NOT ever drink the ethanol you find in a chemistry lab. It is probably poisonous! The liver oxidizes the ethanol people drink. It is turned into acetic acid (vinegar) and acetaldehyde. Together, these make the scent associated with a drunk.
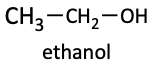
Isopropanol
2-Propanol, or isopropanol, is commonly found in the home. A mixture of 70% isopropanol and 30% water is called rubbing alcohol. A little more than half a cup of isopropanol can kill somebody. When it is oxidized in the liver, it turns into acetone (fingernail polish remover).

Ethylene Glycol and Propylene Glycol
Ethylene glycol and propylene glycol are vicinal diols, or glycols. They are found in antifreeze. They are poisonous. Because antifreeze tastes sweet, we need to be careful if it is spilled on the garage floor. Our pets may enjoy licking it off of the floor, but it can easily kill them. So, be sure to always clean up antifreeze spills!

Glycerol
Glycerol is a clear, thick liquid. Glycerol is normally found in the body. It is made as a product of fat metabolism. It is normally found in animal fats, lard, vegetable oils, etc. Glycerol is water-soluble. It is a lubricant and is used in skin lotions and soaps, and shaving creams. It is found in cake frosting to help prevent the sugar from crystallizing.

Answers
1.







